Charcot-Marie-Tooth Disease Type 1A: A Guide
Understanding CMT1A
Charcot-Marie-Tooth disease type 1A is the most common form of CMT, accounting for about half of people with CMT. CMT1A is commonly referred to as “demyelinating” CMT.
CMT1A, like other forms of CMT1, is inherited in an autosomal dominant fashion. This means you can inherit the disease from either parent if they also have the disease. However, it is important to remember that around 10% of CMT cases are “sporadic,” meaning that rather than being inherited from either of their parents, these individuals had the genetic mutation occur spontaneously during their embryonic development.
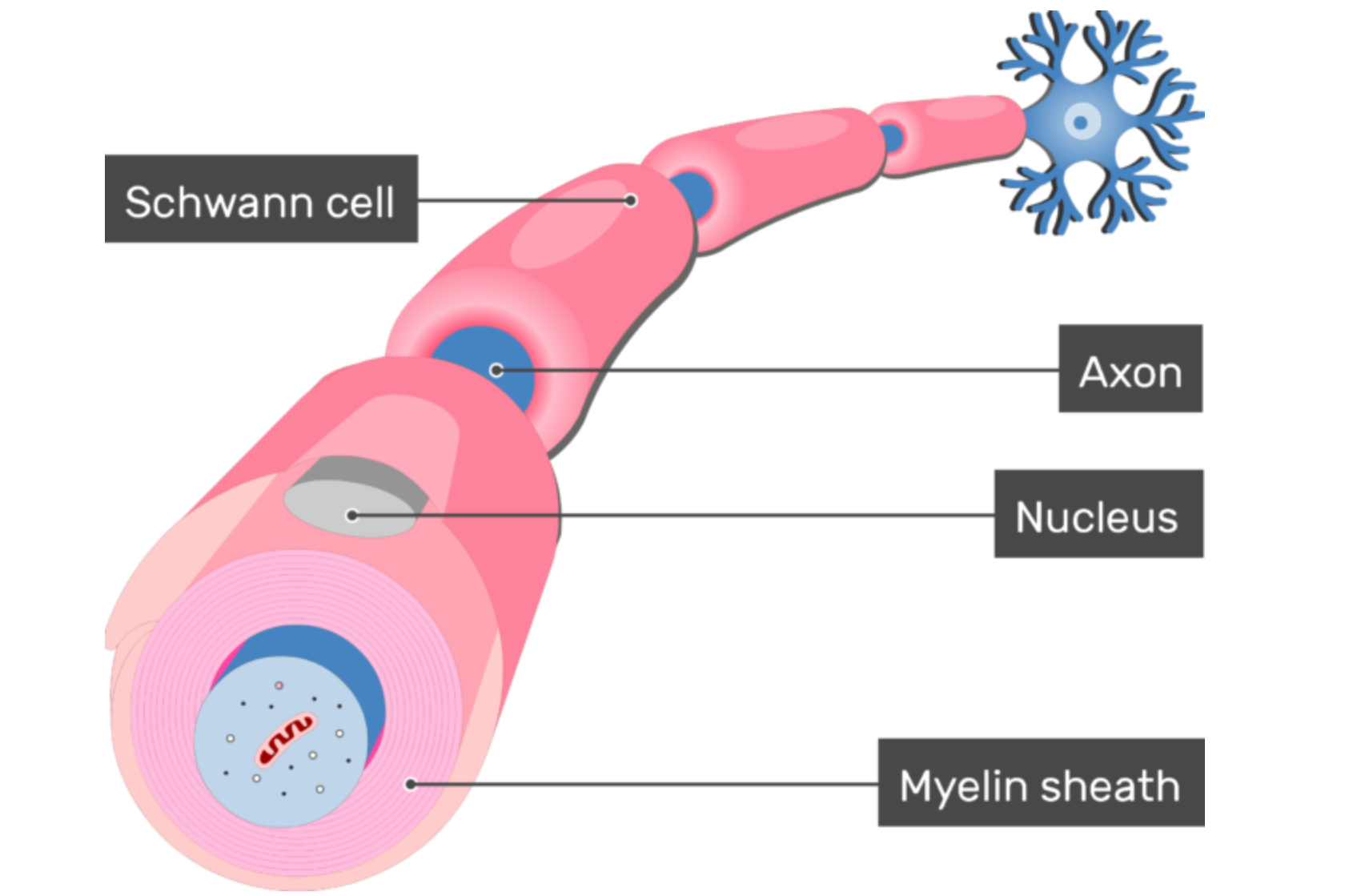
In CMT1A, the part of the peripheral nervous system that is dysfunctional is the myelin — the insulating sheath surrounding the axonal component of nerves, which carries signals to and from the body and central nervous system (brain and spinal cord). In the peripheral nervous system, myelin is produced by cells called Schwann cells. In CMT1A patients, these Schwann cells harbor a duplication of the PMP22 gene (peripheral myelin protein 22 gene).
Causes of CMT1A
The duplication of the PMP22 gene results in a total of three copies of the gene rather than the typical two copies. People without CMT1A inherit one copy of PMP22 from each parent. But in CMT1A, a person inherits one copy of PMP22 from one parent and two copies from the affected parent. This additional copy of the gene results in an over-production of the protein produced by the gene, which is also called PMP22 but usually written without italics. This overabundance of PMP22 is damaging to the Schwann cells producing it and causes malfunctions in the normal myelination process. Exactly how and why this happens is still a mystery to scientists. Because we know that a duplication of the PMP22 gene causes CMT1A, scientists are already pursuing genetic therapies for CMT1A; however, understanding the exact mechanism by which an overabundance of PMP22 protein causes loss of myelin will be a critical breakthrough that opens many new treatment possibilities.
PMP22 is an integral component of the myelin sheath produced by the Schwann cells that insulate the axons of the nerves. In people with CMT1A, the myelin in their peripheral nerves is improperly formed, thinned or even absent. These changes can be seen by microscopic examination within the first year of life and are slowly progressive over the lifespan.
The myelin is essential for the fast and coordinated electrical signaling the human body is accustomed to when communicating with the extremities, especially the muscles. Without consistent myelination, this electrical signaling will slow down and become uncoordinated. Over time, the axons themselves begin to degenerate, and the muscles of the body become weaker and less coordinated, and ultimately begin to deteriorate.
Symptoms of CMT1A
Symptom onset for individuals with CMT1A typically begins by the age of 20, and most often before the age of 10. However, there are still a lot of variabilities, with some people experiencing their first symptoms later in life, and others with identifiable symptoms at birth.
- Pes cavus (or high arch) and hammertoes are the most common symptoms of CMT1A and result from a weakening and wasting of the muscles in the feet.
- Additional common symptoms of CMT1A include difficulty walking or running and a symmetrical weakening and wasting of the muscles in the feet and then the lower legs, with that weakness moving up the legs as the disease progresses. The feet and legs are generally affected first, but as the disease progresses, many individuals will experience a similar weakening of the muscles in the hands which can progress up the arms. This results in diminished dexterity with one’s hands.
- There is also frequently a numbness in the same regions of the body exhibiting muscle deterioration (CMT affects the parts of the peripheral nervous system responsible for both movement and sensation).
- Individuals with CMT1A will often have issues with balance that occur due to damage to their proprioception, or the ability to sense where your body’s parts are in relationship to one another and how they are moving in space.
Read more about CMT signs and symptoms
Treatments for CMT1A
There are currently no FDA-approved treatments for CMT1A. However, the development of drugs to treat CMT1A has been an active area of research interest. One common therapeutic target is to reduce the amount of PMP22 protein in the Schwann cells. Previous attempts to do this were largely unsuccessful following large-scale clinical trials. However, new technology and new drugs are being tested in clinical trials to achieve the same goal, hopefully with greater success. Find ongoing CMT1A clinical trials here.
The CMT Research Foundation is funding multiple projects focused on developing CMT1A treatments using various gene therapy technologies.
Li, J., Parker, B., Martyn, C., Natarajan, C., & Guo, J. (2013). The PMP22 gene and its related diseases. Molecular Neurobiology, 47(2), 673–698. https://doi.org/10.1007/s12035-012-8370-x
Morena, J., Gupta, A., & Hoyle, J. C. (2019). Charcot-Marie-Tooth: From molecules to therapy. International Journal of Molecular Sciences, 20(14). https://doi.org/10.3390/ijms20143419
Saporta, A. S. D., Sottile, S. L., Miller, L. J., Feely, S. M. E., Siskind, C. E., & Shy, M. E. (2011). Charcot-Marie-Tooth disease subtypes and genetic testing strategies. Annals of Neurology, 69(1), 22–33. https://doi.org/10.1002/ana.22166
Van Paassen, B. W., Van Der Kooi, A. J., Van Spaendonck-Zwarts, K. Y., Verhamme, C., Baas, F., & De Visser, M. (2014). PMP22 related neuropathies: Charcot-Marie-Tooth disease type 1A and hereditary neuropathy with liability to pressure palsies. Orphanet Journal of Rare Diseases, 9(1), 1–15. https://doi.org/10.1186/1750-1172-9-38

