CMT Drug Development Process and Therapy Pipeline
Therapies Under Development in Industry
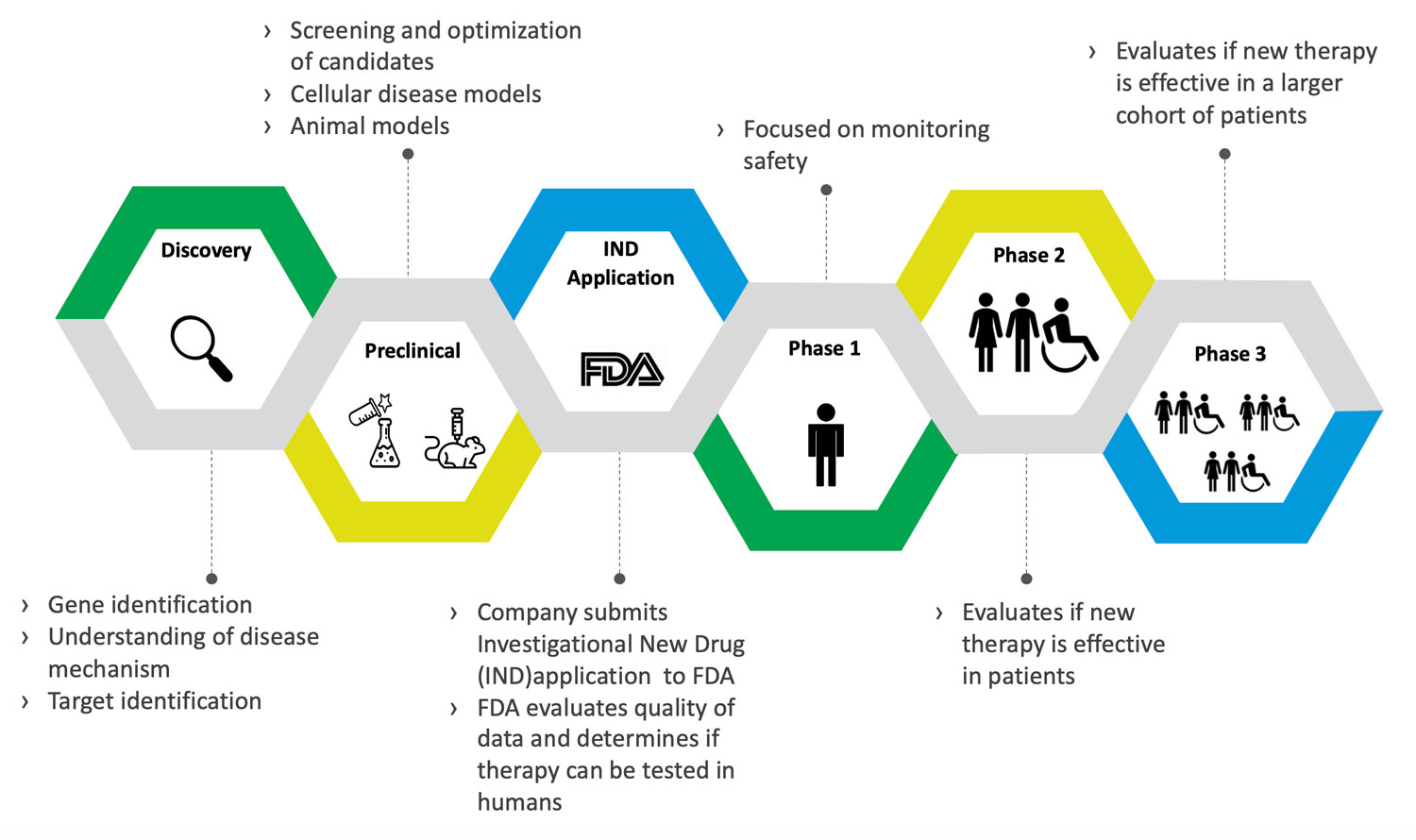
Therapy Pipeline
PMO-ASO
*Funded by CMTRF
**CMTRF funded underlying technology
***CMTRF funded drug manufacturing
Updated 7/2025
Today, available treatments for CMT are designed to help people effectively manage disease symptoms. Drugs may be used to lessen pain, and orthopedic surgery to correct joint deformities. Physical and occupational therapy often incorporate strength training, muscle and ligament stretching and exercise to help with muscle weakness and pain. In addition, a range of medical devices including braces, walkers and wheelchairs are used to help maintain mobility, assisted ventilation can help with breathing issues and hearing aids help compensate for hearing loss.
However, with the identification over the past several decades of more than 100 different gene mutations underlying the various forms of CMT, scientists now have a broad range of targets at which to aim therapies that can modify the course of the disease.
CMTRF is working hard today to make good on our promise to deliver safe and effective treatments to people living with all forms of CMT. Our expectation is that the treatments of tomorrow will do more than manage symptoms. Tomorrow’s treatments, under development and testing now, will aim to modify the course of the disease whether by targeting a particular function, or changing a person’s genetic makeup.
